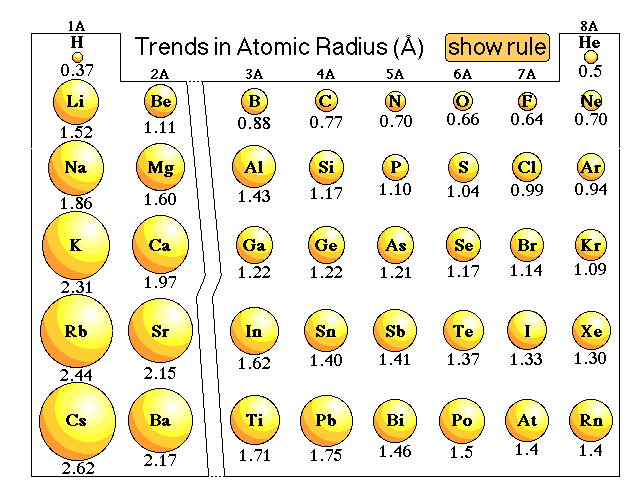
Sodium is obtained commercially by electrolysis of molten sodium chloride. Trends in atomic radius in Periods 2 and 3. The D lines of sodium are among the most prominent in the solar spectrum. The truth is that atomic weights have changed as a function of time. In addition chemistry and technical terms are linked to their definitions in the site's chemistry and environmental dictionary. Sodium has a larger atomic radius and is more metallic When an atom loses one one or more electrons,this atom becomes a Positive ion with a radius smaller than the radius of this atom … Atomic and ionic radii are found by measuring the distances between atoms and ions in chemical compounds. The atomic radius of a chemical element is the distance from the centre of the nucleus to the outermost shell of the electron. Potassium (K) has an atomic radius of 227. Therefore the atomic volume of sodium is 11.763. It has more electrons too, which balance out that charge, but there are many more electrons in the shell that is farthest away. Isotopes: Sodium has 16 isotopes whose half-lives are known, with mass numbers 20 to 35. As there are no physical existence of orbital in atoms, it is difficult to measure the atomic radius.

Assuming atoms and molecules are spheres, the radius is the cube root of 11.763 divided by (pi x 4/3).

495.8, 4562, 6910.3, 9543, 13354, 16613, 20117, 25496, 28932, 141362 kJ/mol Comparing carbon (C) with an atomic number of 6 and fluorine (F) with an atomic number of 9, we can tell that, based on atomic radius trends, a carbon atom will have a larger radius than a fluorine atom since the three additional protons the fluorine has will pull its electrons closer to the nucleus and shrink the fluorine's radius.
